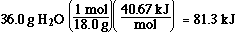How much heat is required to change 36.0 g of H2O(l) at 100 ûC
to 36.0 g of H2O(g) at 100 ûC?
I got 81.3 kJ. Is that what you got? If not go here to see how I solved the
problem.
Solution:
The conversion requires changing from the liquid phase to the solid phase.
This is vaporization. So all we need is the enthalpy of vaporization and the
mol of water.